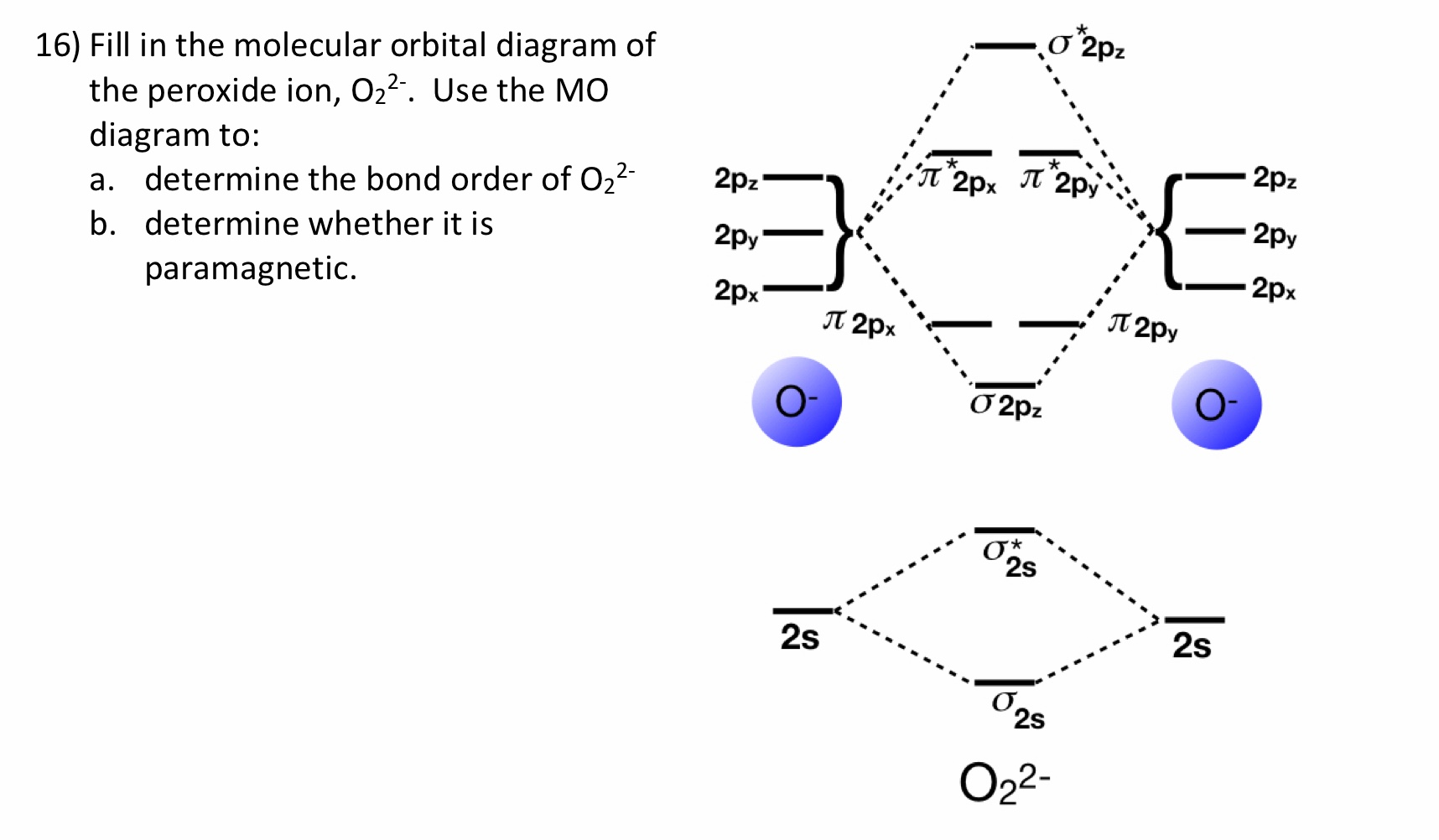
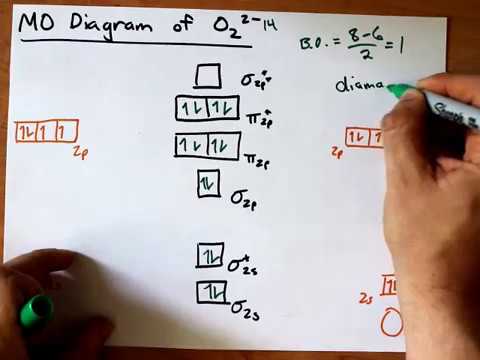
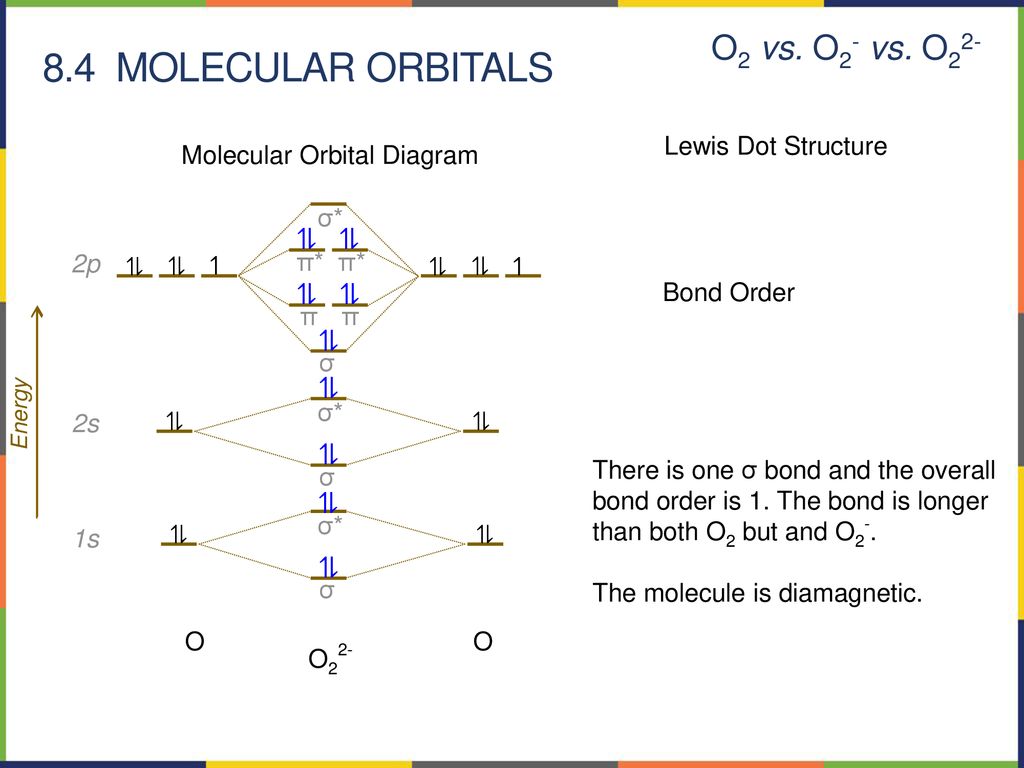
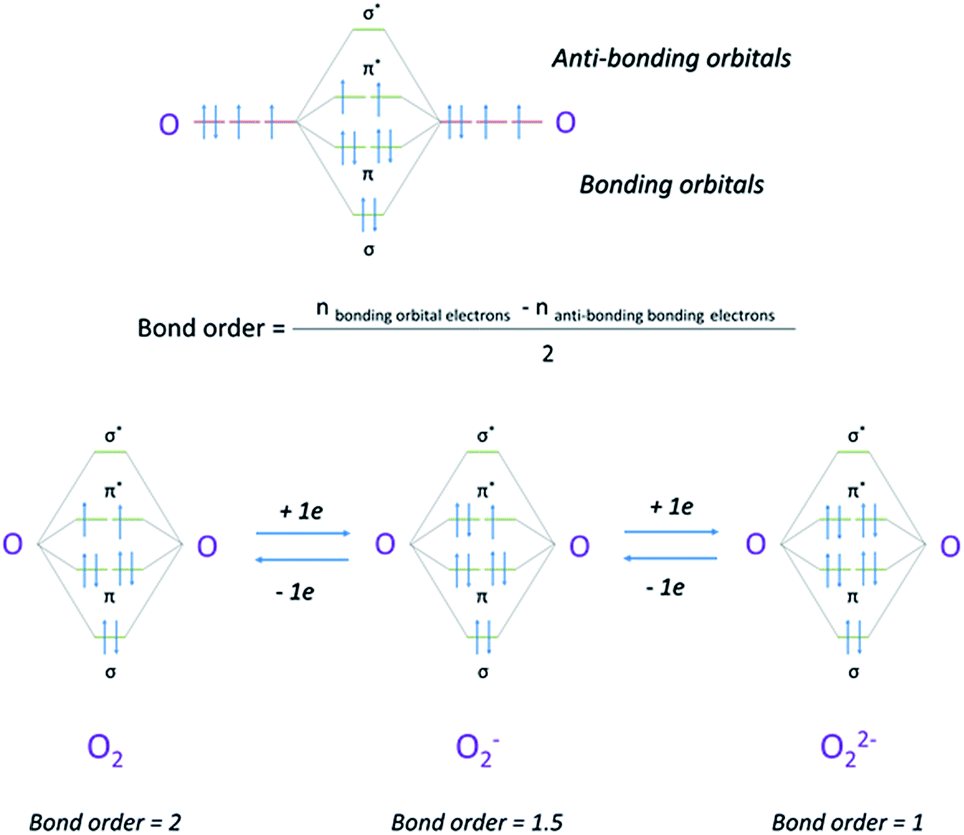
39 o22- molecular orbital diagram
Ans: The stabilities of these can be best explained using Molecular orbital theory. ... Atomic orbitals of oxygen combine to form molecular ... 5.7 a. The energy level diagram for NO is on the right. The odd electron is in a π2p* orbital. b ...
Draw the MO diagram for O22- and identify the following: Bond order, diamagnetic or paramagnetic, homo and lumo. ... Hey there! We will begin by drawing the ...

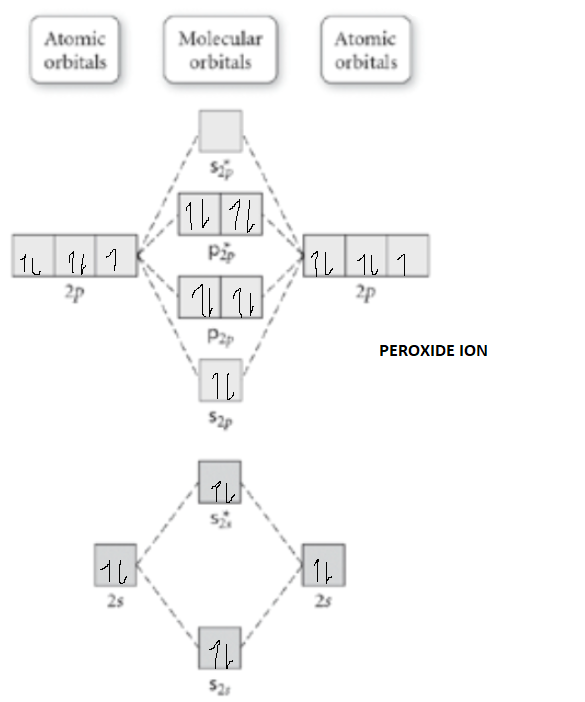
O22- molecular orbital diagram
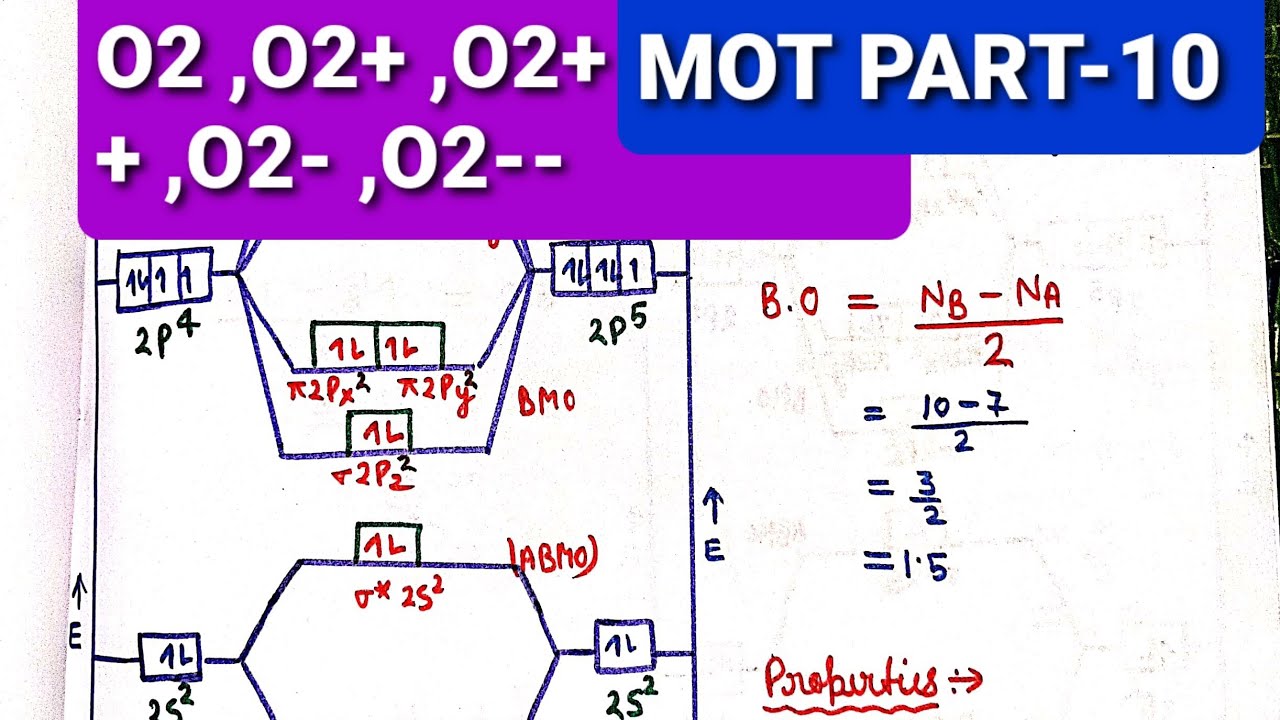
Answer to: Molecular Orbital Theory -- Homodiatomics use the molecular orbital model to fully describe the bonding in O2+, O2, O2-, and O22-.... It is sigma2s(2)sigma2s*(2)sigma2p(2)pi2p(4)pi2p*(4)Bond order 1. It is stable. In fact, it's the perioxide ion. How To make molecular Orbital diagramhttps://www.youtube.com/watch?v=UYC-ndQ6Lww&t=6s.
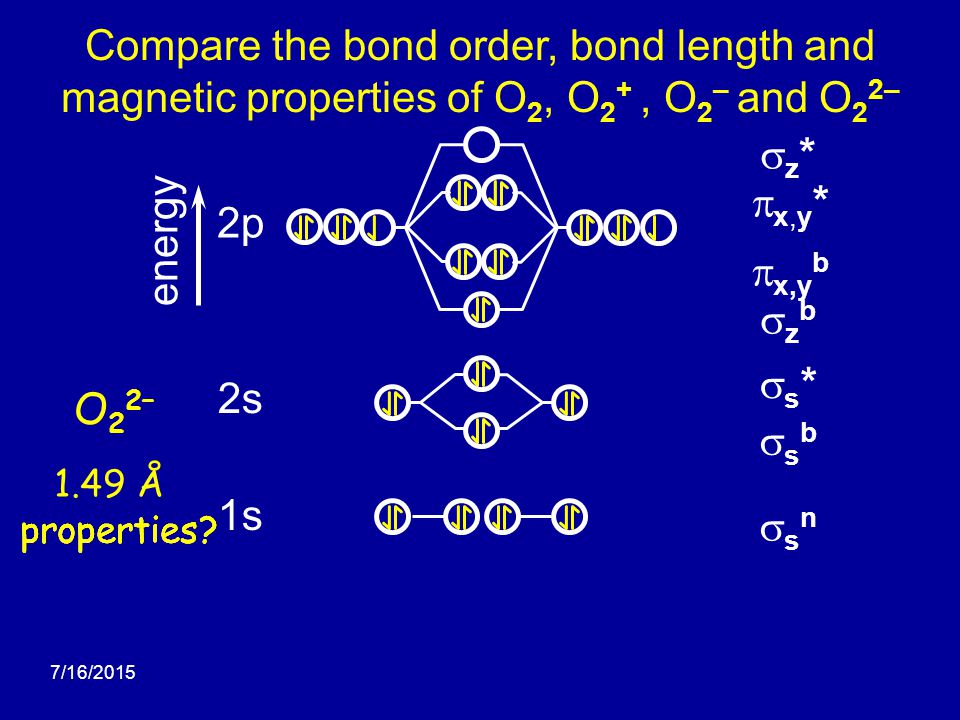
O22- molecular orbital diagram. This is the peroxide ion, O2(2-), so you KNOW it's going to be stable.It has a bond order of 1, which also makes sense. Can you draw good dot structures that correspond to each of these ions or molecules? Draw a molecular orbital diagram lor Arz*. This ion has been observed in ... The first photo is straight from a 2006 edition Pearson general chemistry textbook, and it shows you what the molecular orbital (MO) diagram for O2 is. Hint: First draw a molecular orbital diagram (MOT) where the atomic orbitals combine to form molecular orbitals. The total electrons associated with the ...
How To make molecular Orbital diagramhttps://www.youtube.com/watch?v=UYC-ndQ6Lww&t=6s. It is sigma2s(2)sigma2s*(2)sigma2p(2)pi2p(4)pi2p*(4)Bond order 1. It is stable. In fact, it's the perioxide ion. Answer to: Molecular Orbital Theory -- Homodiatomics use the molecular orbital model to fully describe the bonding in O2+, O2, O2-, and O22-....

Solved Use Molecular Orbital Theory Write A Molecular Orbital Diagram Calculate Bond Order And Predict Whether Or Not Each Ion Exists In A Relatively Stable Form For The Following Two Molecular Ions

Based On The Mo Diagrams For O 2 O 2 And O 2 Answer The Following 1 Is O 2 Paramagnetic Or Diamagnetic 2 Which Will Have The Shortest Bond Length 3 Which Will Have The
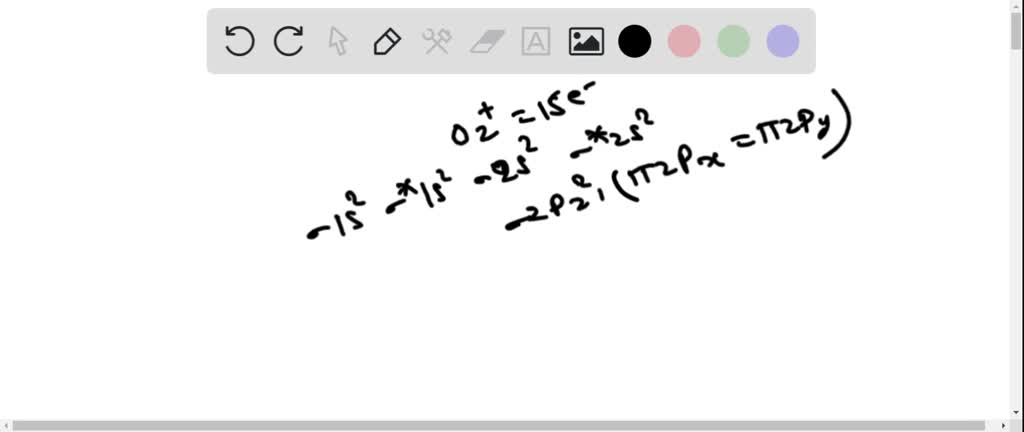
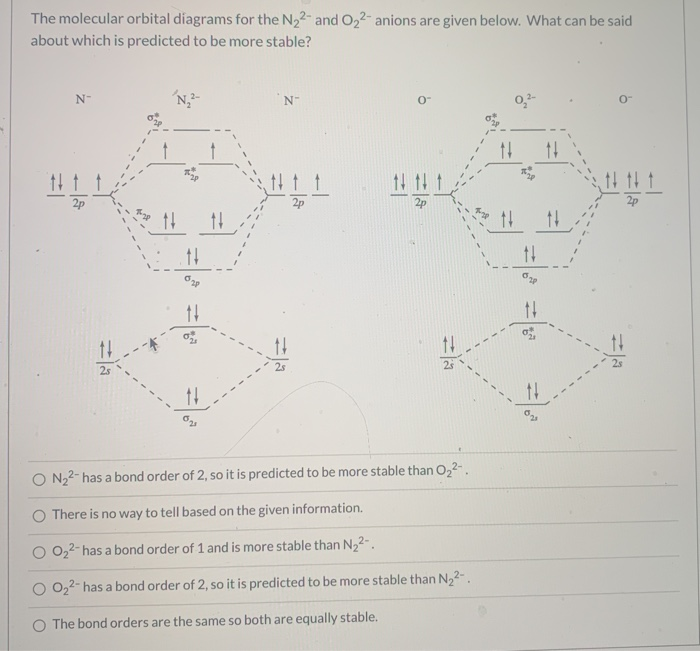
Solved According To The Molecular Orbital Energy Level Diagram Which Of The Following Statements Are True About O22 A The Oxygen Oxygen Bond Order Is 1 B The Ion Has An Odd Number Of Electrons

Draw The Molecular Orbital Diagram Shown To Determine Which Of The Following Is Paramagnetic N22 B22 Homeworklib
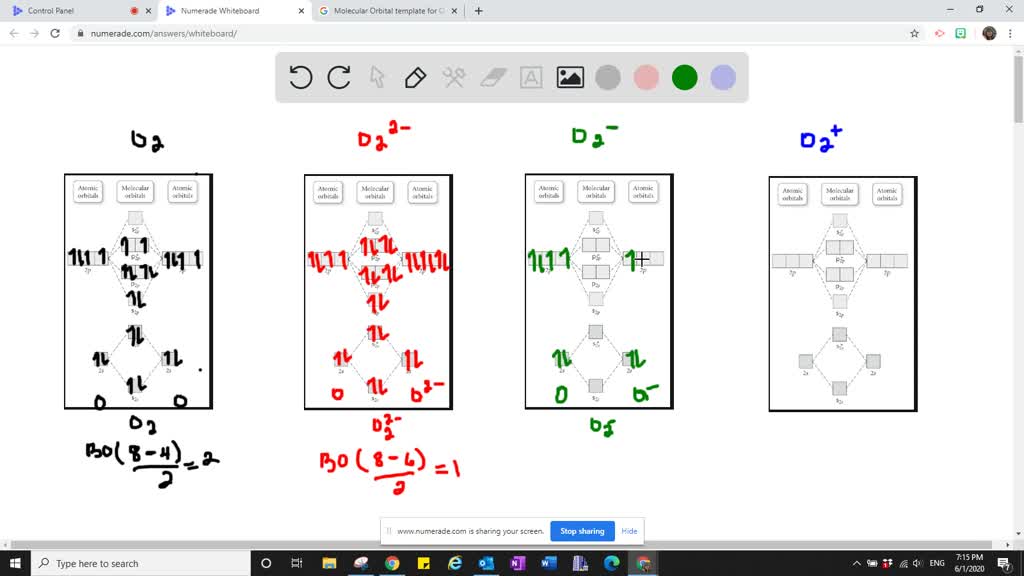
Solved The Superoxide Ion O2 Is A Reactive Species That May Play A Role In The Chemistry Of Aging Use A Molecular Orbital Diagram To Determine The Bond Order Of The Superoxide Ion
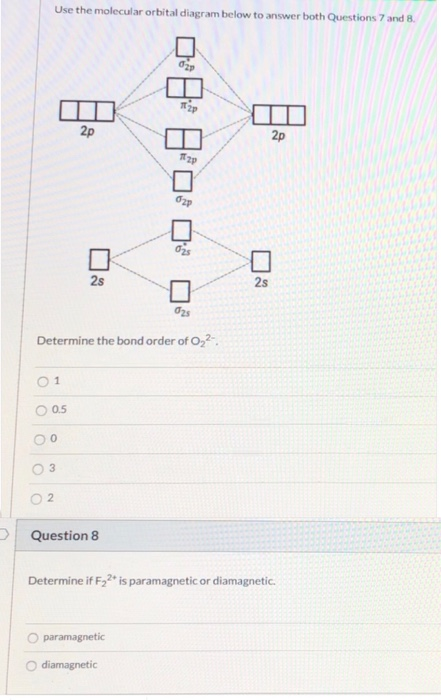
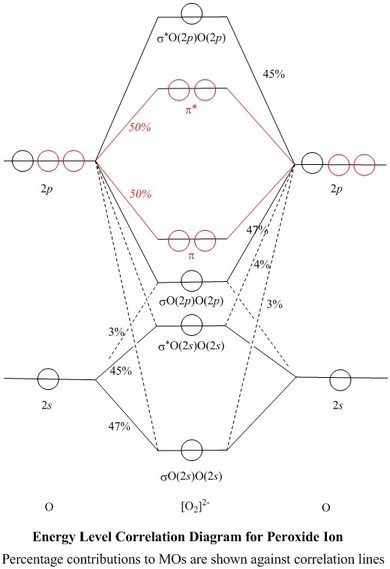
















0 Response to "39 o22- molecular orbital diagram"
Post a Comment