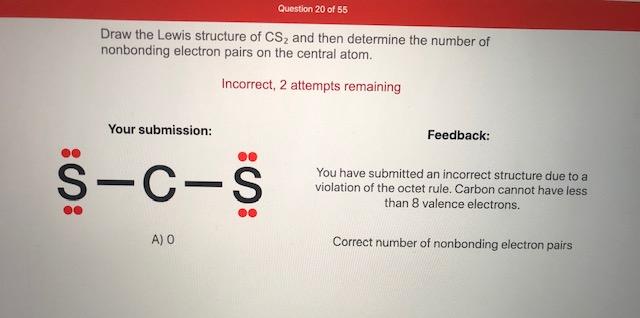
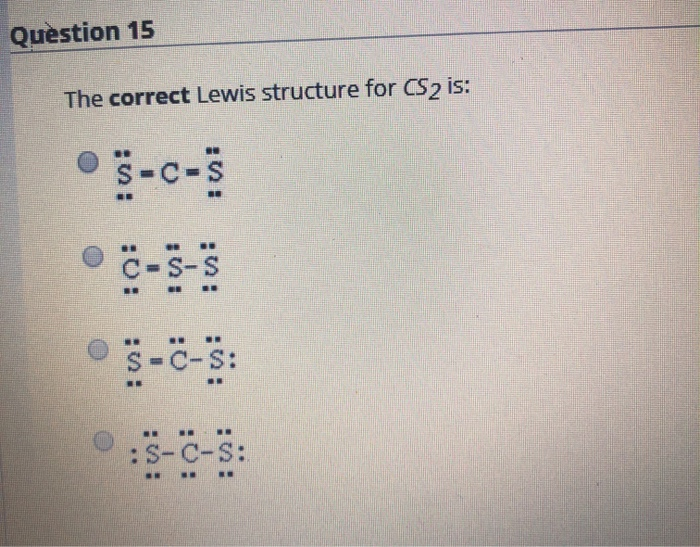
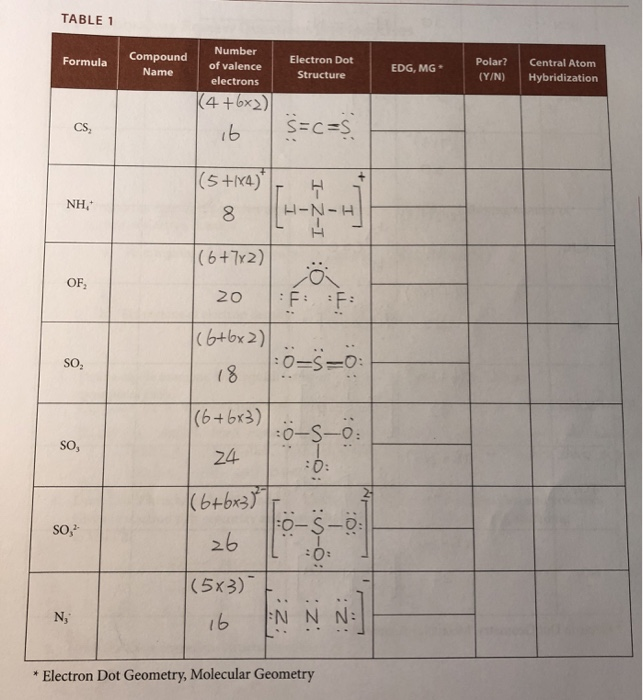
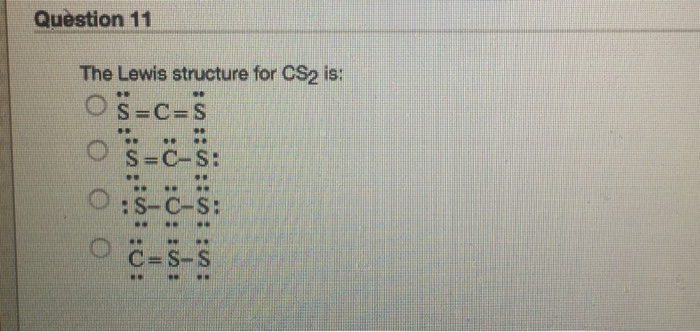
38 lewis dot diagram for cs2
A step-by-step explanation of how to write the Lewis Dot Structure for CS2 (Carbon DiSulfide). For the CS2 Lewis structure, calculate the total number of valence electrons for the CS2 molecule. After determining how many valence electrons there are in CS2, place them around the central atom to complete the octets. Let's do the Lewis structure for CS2. On the periodic table, Carbon's in group 4, sometimes called 14, so it has 4 valence electrons. Sulfur in group 6 or 16, it has 6. We have two Sulfurs so let's multiply that by 2. Four plus 12: 16 valence electrons. Let's draw it.
The lewis dot structure for CS2 also predicts many of the properties of the molecule. Due to the presence of large sulfide atoms over for comparison oxygen atoms in CO2 the molecule has a greater capacity for temporary london dispersion forces. As a result there can be more induced dipoles which increases the solubility of CS2.

Lewis dot diagram for cs2
To draw the Lewis structure of, we need to calculate the total number of valence electrons.This is done by taking the sum of valence electrons from individual atoms. The outer-shell electronic configuration of is and that of S is.. Number of valence electrons in = 4. Number of valence electrons in = 6. Here we have two atoms and the total number of valence electrons in CS2 Lewis Structure. Answer: CS2 Lewis structure (carbon disulfide electron dot structure) is that type of diagram where we show the total 16 valence electrons of CS2 as dots , or dots and dashes (-).In Lewis structure,it is common that a bonding pair of two electrons can be shown by dash (-) or dots ( ) but a lone pair of two electrons is ... This would also satisfy the preference of S to form 2 bonds. The incomplete Lewis structure of CS2 is. A double bond corresponds to four electrons, so currently ...1 answer · Top answer: We’re being asked to draw a Lewis structure for CS2.To answer this problem, we must:Step 1. Find the center atom of this compound.Step 2. Determine the ...
Lewis dot diagram for cs2. A step-by-step explanation of how to draw the CS2 Lewis Dot Structure (Carbon disulfide).For the CS2 structure use the periodic table to find the total numbe... CS2 is sometimes used to fumigate railroad cars and grain elevators. CS2 is named Carbon Disulfide. There are 16 valence electrons available for the Lewis ...12 Aug 2013 · Uploaded by Wayne Breslyn Lewis Structure is one of the key terminologies to understand the chemical bonding of a molecule since it represents the molecular structure. It depends on the octet rule concept and is an extension of the electron dot diagram. Thus, to have a comprehensive idea about CS2 Lewis Structure, let us go through each step clearly and systematically. In Lewis structure of CS2 molecule, there are 16 valence electrons, out of which four valence electrons are of Carbon, and six valence ...15 Feb 2021 · Uploaded by Geometry of Molecules
In the CS2 Lewis structure diagram, the carbon atom can be the center atom of the molecule. As a result, central carbon in the CS2 Lewis structure, with all two sulfur atoms arranged in a linear geometry. Add valence electron around the sulfur atom, as given in the figure. Step-3: Lewis dot Structure for CS2 generated from step-1 and step-2 A video explanation of how to draw the Lewis Dot Structure for Carbon Disulfide, along with information about the compound including Formal Charges, Polarity... This would also satisfy the preference of S to form 2 bonds. The incomplete Lewis structure of CS2 is. A double bond corresponds to four electrons, so currently ...1 answer · Top answer: We’re being asked to draw a Lewis structure for CS2.To answer this problem, we must:Step 1. Find the center atom of this compound.Step 2. Determine the ... CS2 Lewis Structure. Answer: CS2 Lewis structure (carbon disulfide electron dot structure) is that type of diagram where we show the total 16 valence electrons of CS2 as dots , or dots and dashes (-).In Lewis structure,it is common that a bonding pair of two electrons can be shown by dash (-) or dots ( ) but a lone pair of two electrons is ...
To draw the Lewis structure of, we need to calculate the total number of valence electrons.This is done by taking the sum of valence electrons from individual atoms. The outer-shell electronic configuration of is and that of S is.. Number of valence electrons in = 4. Number of valence electrons in = 6. Here we have two atoms and the total number of valence electrons in


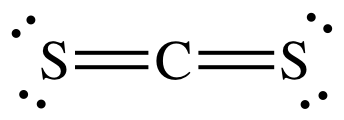


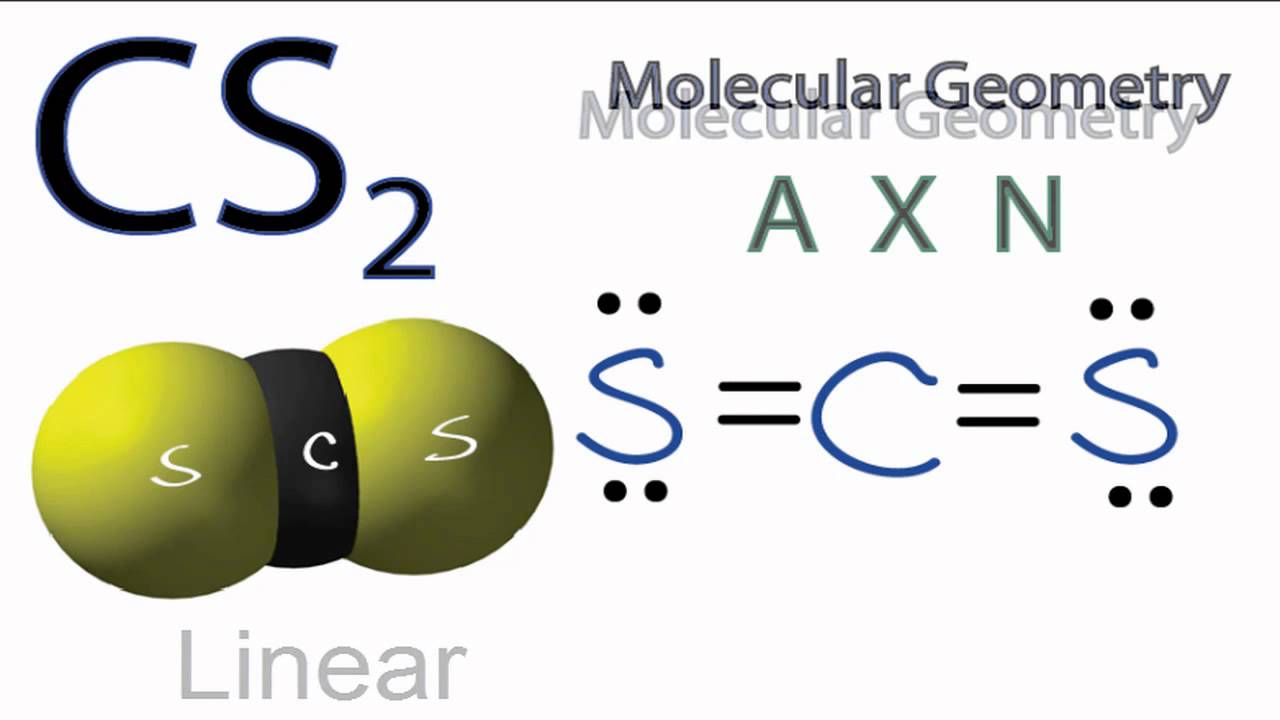

















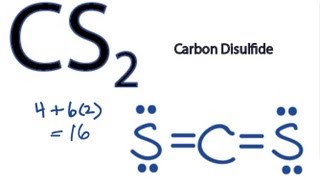


0 Response to "38 lewis dot diagram for cs2"
Post a Comment